SOLVED: Calculate the standard cell potential of the following cell at 25'C: Pb(s) Pb2+ (aq) || Cu? + (aq) Cu(s) Standard Electrode (Reduction) Potentials in Aqueous Solution at 258C Cathode (Reduction) Half-Reaction

Standard Cell Potential: Calculations, Electron Flow & Feasibility (5.4.2) | CIE A Level Chemistry Revision Notes 2022 | Save My Exams

Question Video: Calculating the Standard Cell Potential for a Magnesium/Silver Galvanic Cell | Nagwa

Given standard electrode potentials, Fe^2 + 2e^-→ Fe, E^∘ = - 0.440 V Fe^3 + + 3e^-→ Fe, E^∘ = - 0.036 V The standard electrode potential (E^∘) for Fe^3 + + e^-→ Fe^2 + is:

Calculate the standard electrode potential of the Ni^(2+)//Ni electrode , if the cell potential ... - YouTube

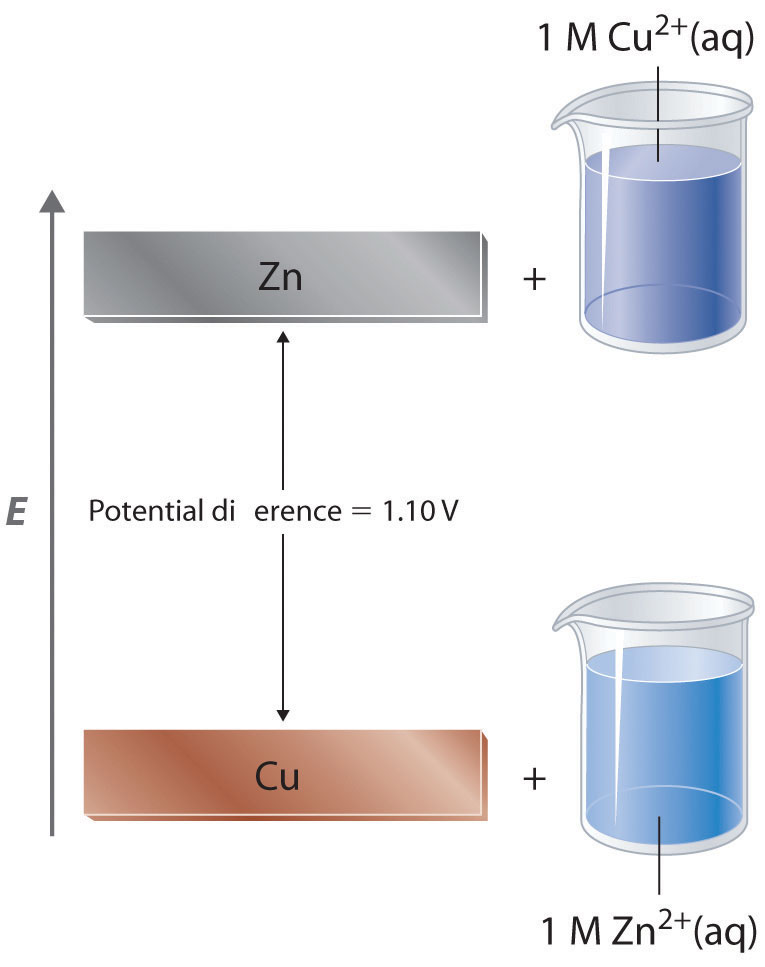
The standard electrode potential for Daniell cell is 1.1V. Calculate the standard Gibbs energy for.. - YouTube
2 The standard half reduction potential of Ag+|Ag is 0.79V is 25^° C. Given the experimental value Ksp=1.5 10* 10 for AgCl, calculate the standard half cell reduction potential for the Ag|AgCl

Calculate the standard electrode potential of Ni2+/Ni electrode if |Class 12 CHEMISTRY | Doubtnut - YouTube