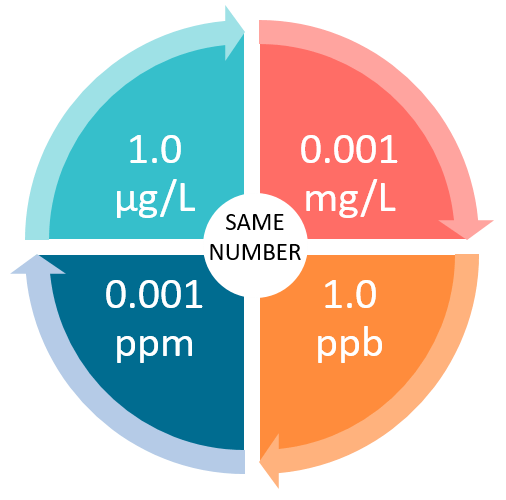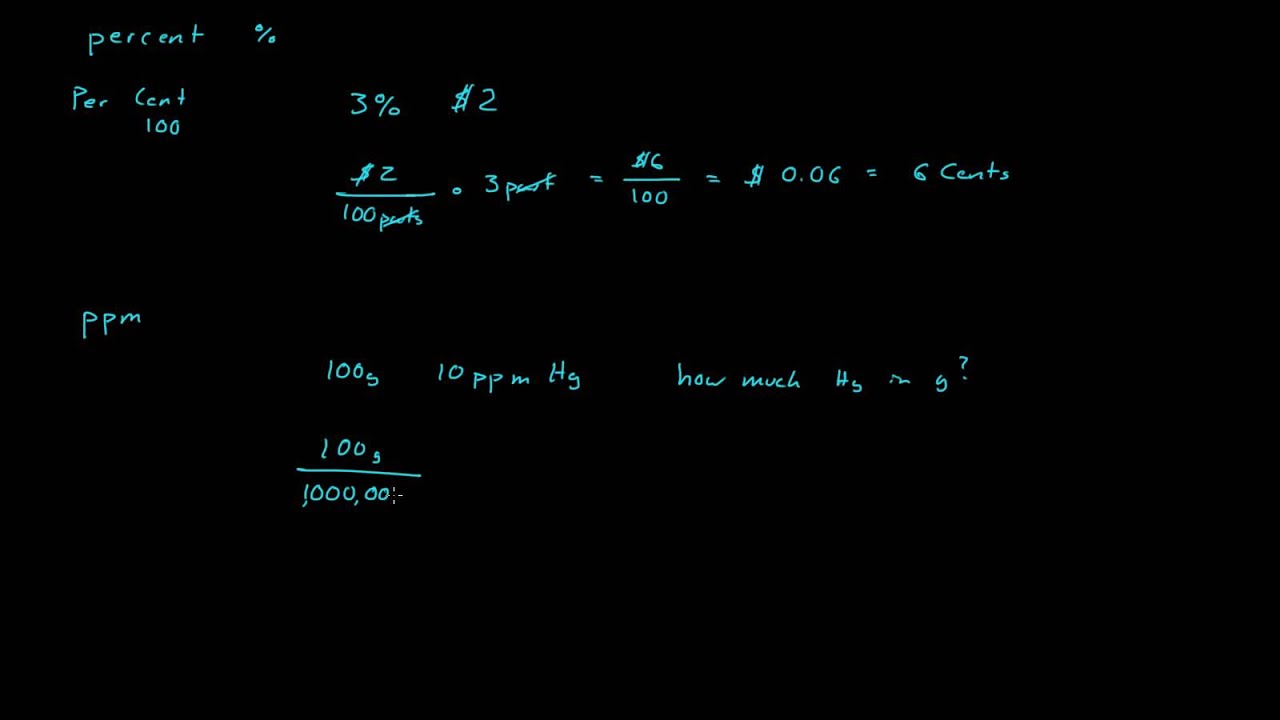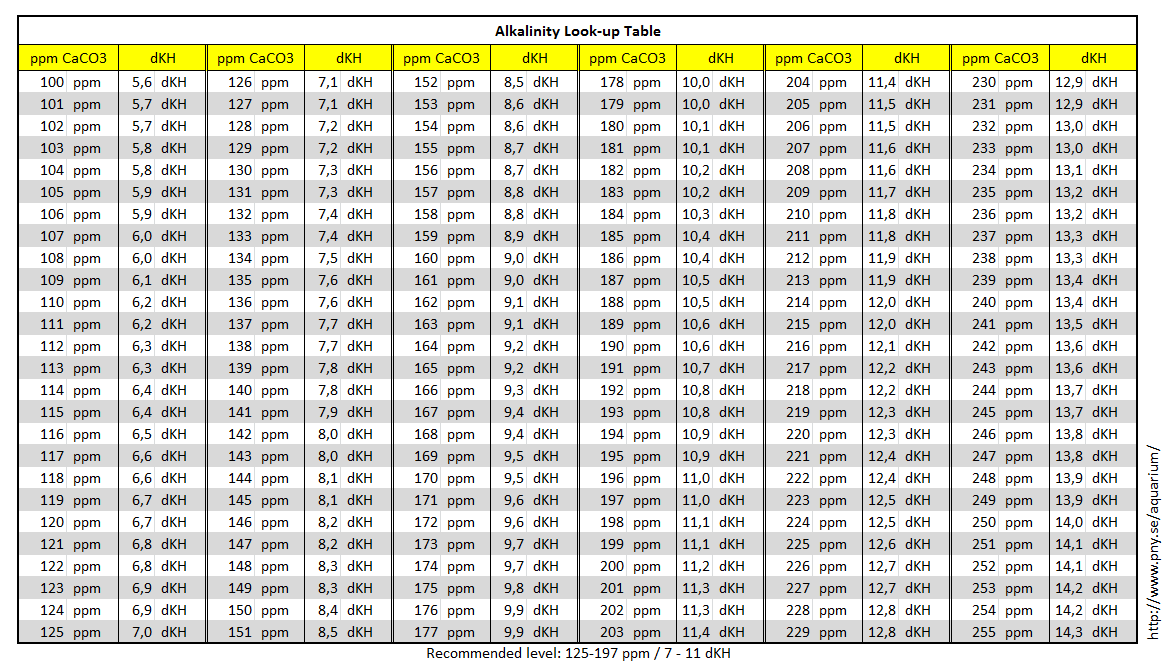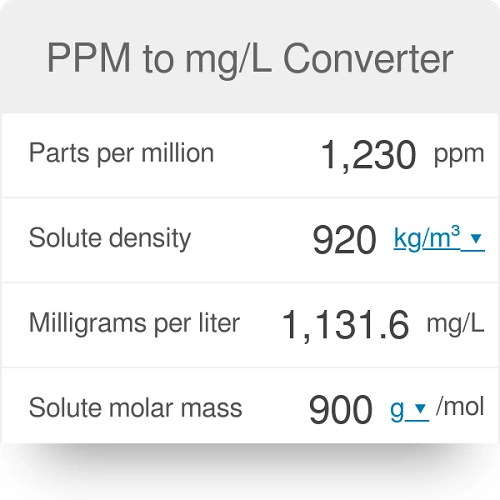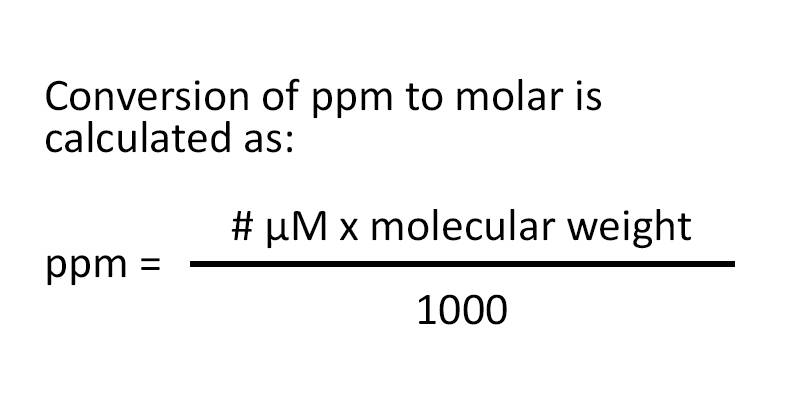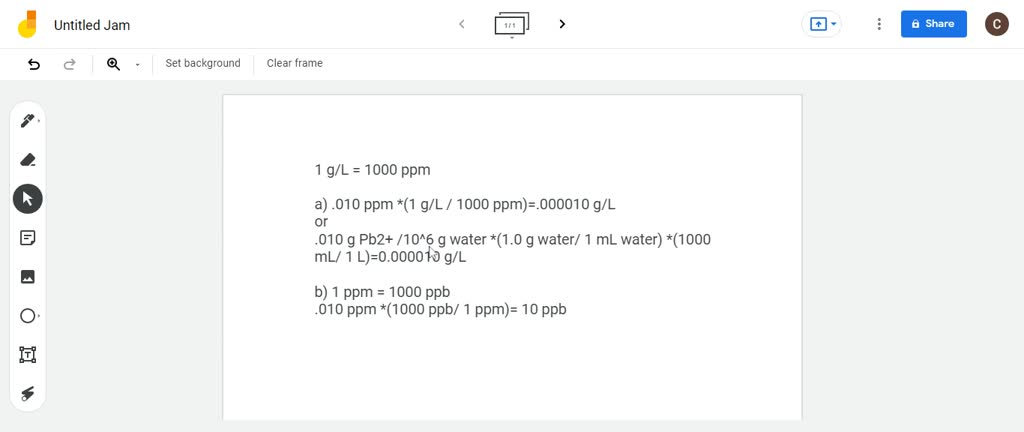
SOLVED:A sample of water contains 0.010 ppm lead ions, Pb^2+. (a) Calculate the mass of lead ions per liter in this solution. (Assume the density of the water solution is 1.0 g /

Converting Units of Concentration in General Chemistry: M, m, ppm, ppb, mass %, mole fraction - YouTube
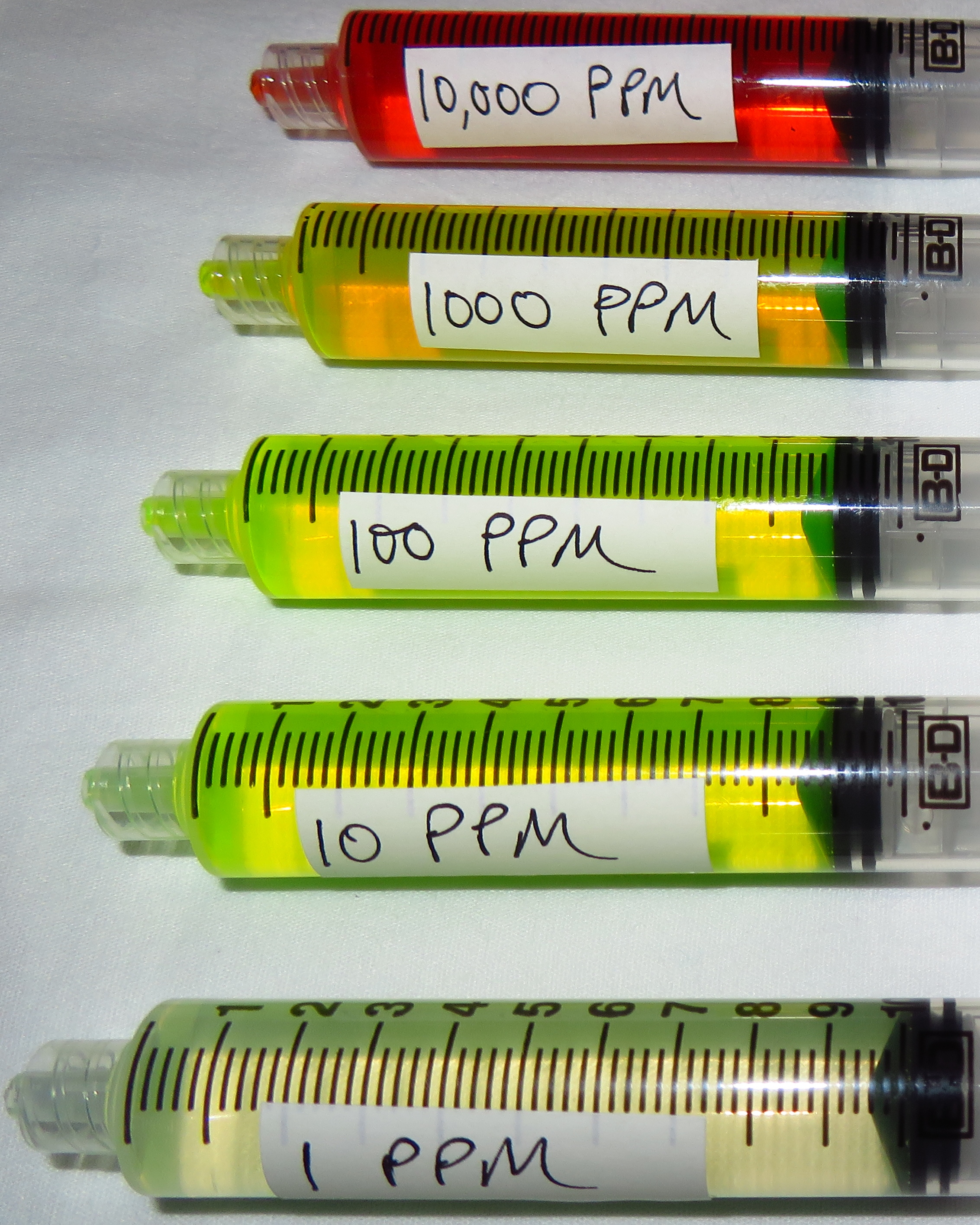
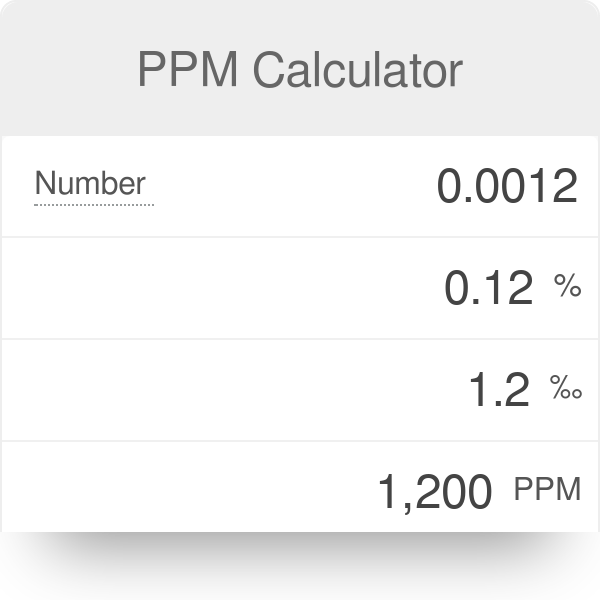
Measurement Units and Concentration Analogies 1 ppm = 1,000 ppb = 1,000,000 ppt 0.001 ppm = 1ppb = 1,000 ppt
meteorology - How do I convert kg·kg⁻¹ to ppbV (parts per billion volume)? - Earth Science Stack Exchange