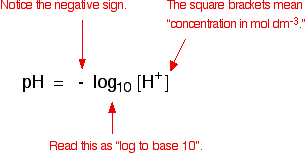
![Calculating pH and pOH. pH pH = - log [H + ] [H + ] = the hydrogen ion concentration pH: “potential of hydrogen” - A way of expressing the hydrogen ion. - ppt download Calculating pH and pOH. pH pH = - log [H + ] [H + ] = the hydrogen ion concentration pH: “potential of hydrogen” - A way of expressing the hydrogen ion. - ppt download](https://images.slideplayer.com/17/5302527/slides/slide_3.jpg)
Calculating pH and pOH. pH pH = - log [H + ] [H + ] = the hydrogen ion concentration pH: “potential of hydrogen” - A way of expressing the hydrogen ion. - ppt download

Calculating Hydronium ion concentration from pH (and identifying molecules based on pH) : r/chemhelp
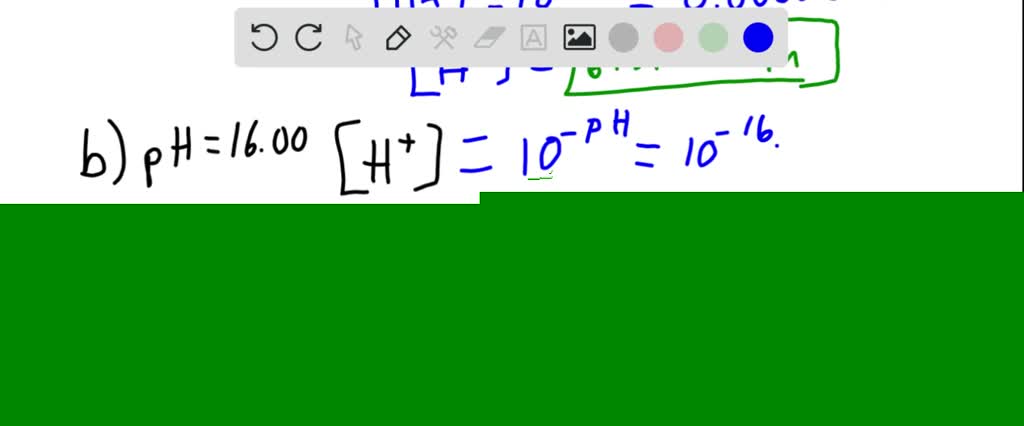








:max_bytes(150000):strip_icc()/how-to-calculate-ph-quick-review-606089_final-165915b0177b4f6e82843f25097f51df.png)




![Calculate [H+] from pH - YouTube Calculate [H+] from pH - YouTube](https://i.ytimg.com/vi/yE4M2Uuh-h4/sddefault.jpg)

