
SOLVED: 5. How many grams of NaCl are required to 1.50 liters of a 0.100 M solution? a. 0.150 grams b. 8.77 grams c. 0.00257 grams d. 0.114 grams e. None of

Intro to Quantities Review Problems These are the calculations you should be able to perform: Sum of molar mass for a compound Convert mass mole Convert. - ppt download

Molarity Dilution Problems Solution Stoichiometry Grams, Moles, Liters Volume Calculations Chemistry - YouTube

The What Is A Mole? A Conversion Factor We Use In Chemistry To Make It Easier To Talk About A Very Large Amount Of Particles Of Elements Or Ppt | xn--90absbknhbvge.xn--p1ai:443
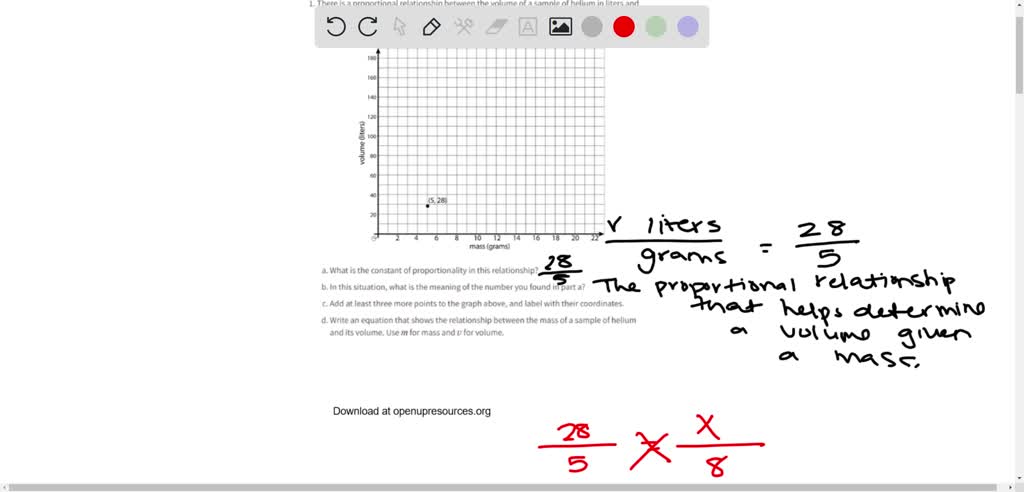
SOLVED:There is a proportional relationship between the volume of a sample of helium in liters and the mass of that sample in grams. If the mass of a sample is 5 grams,




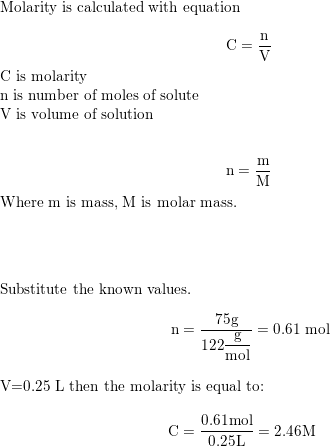










![Learn Metric Units & Unit Conversions (Meters, Liters, Grams, & more) - [5-8-1] - YouTube Learn Metric Units & Unit Conversions (Meters, Liters, Grams, & more) - [5-8-1] - YouTube](https://i.ytimg.com/vi/T8WZU1QqSgI/maxresdefault.jpg)

