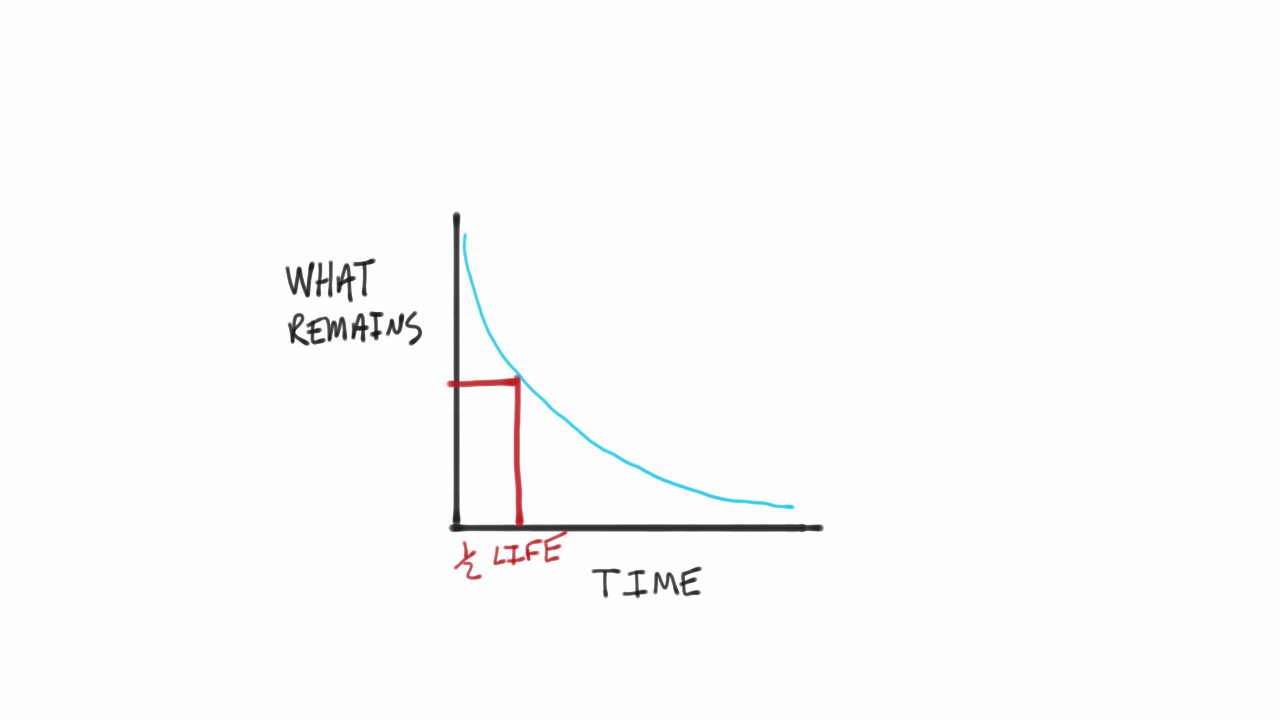
Define the term half-life period and decay constant of a radioactive substance. Write their `S.I` - YouTube

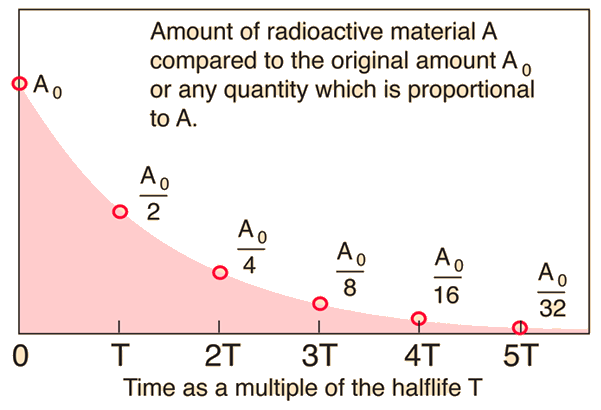
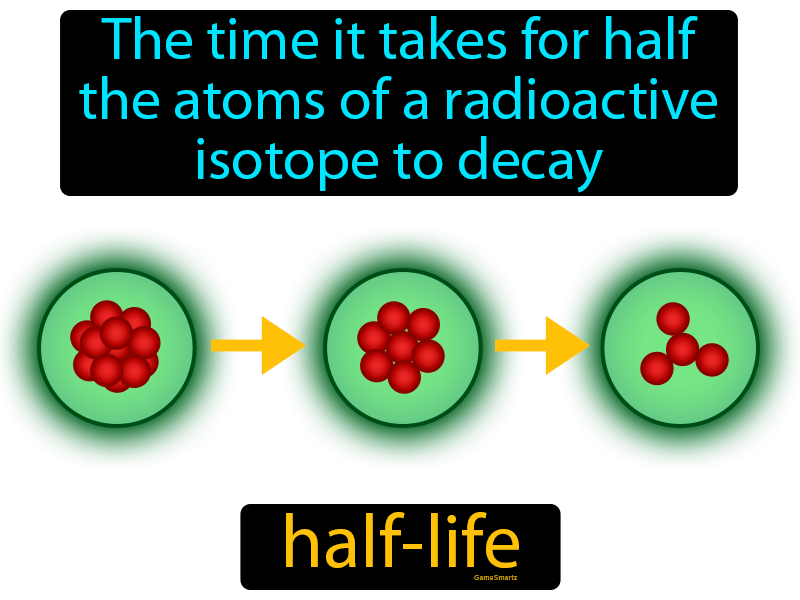
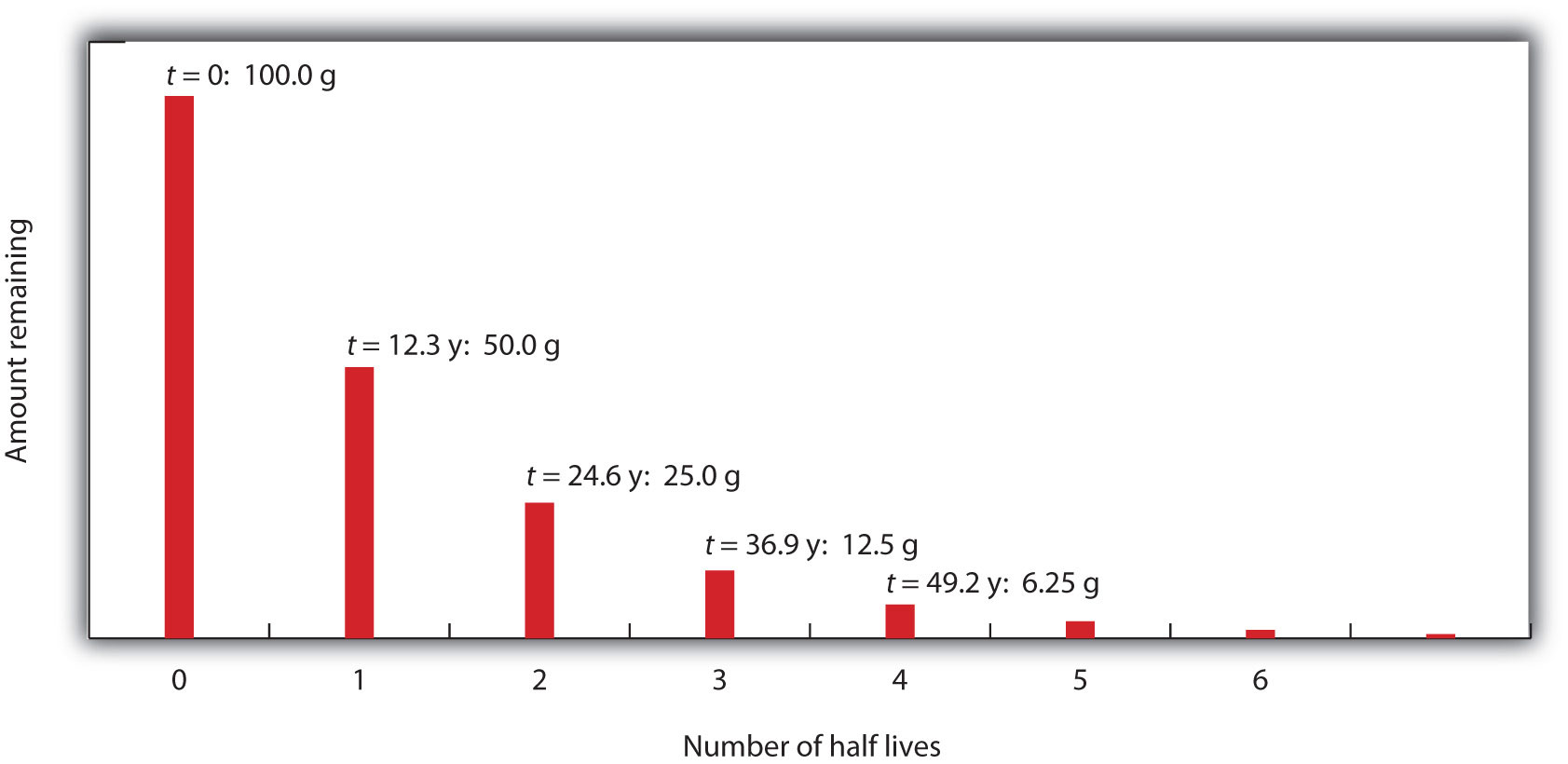
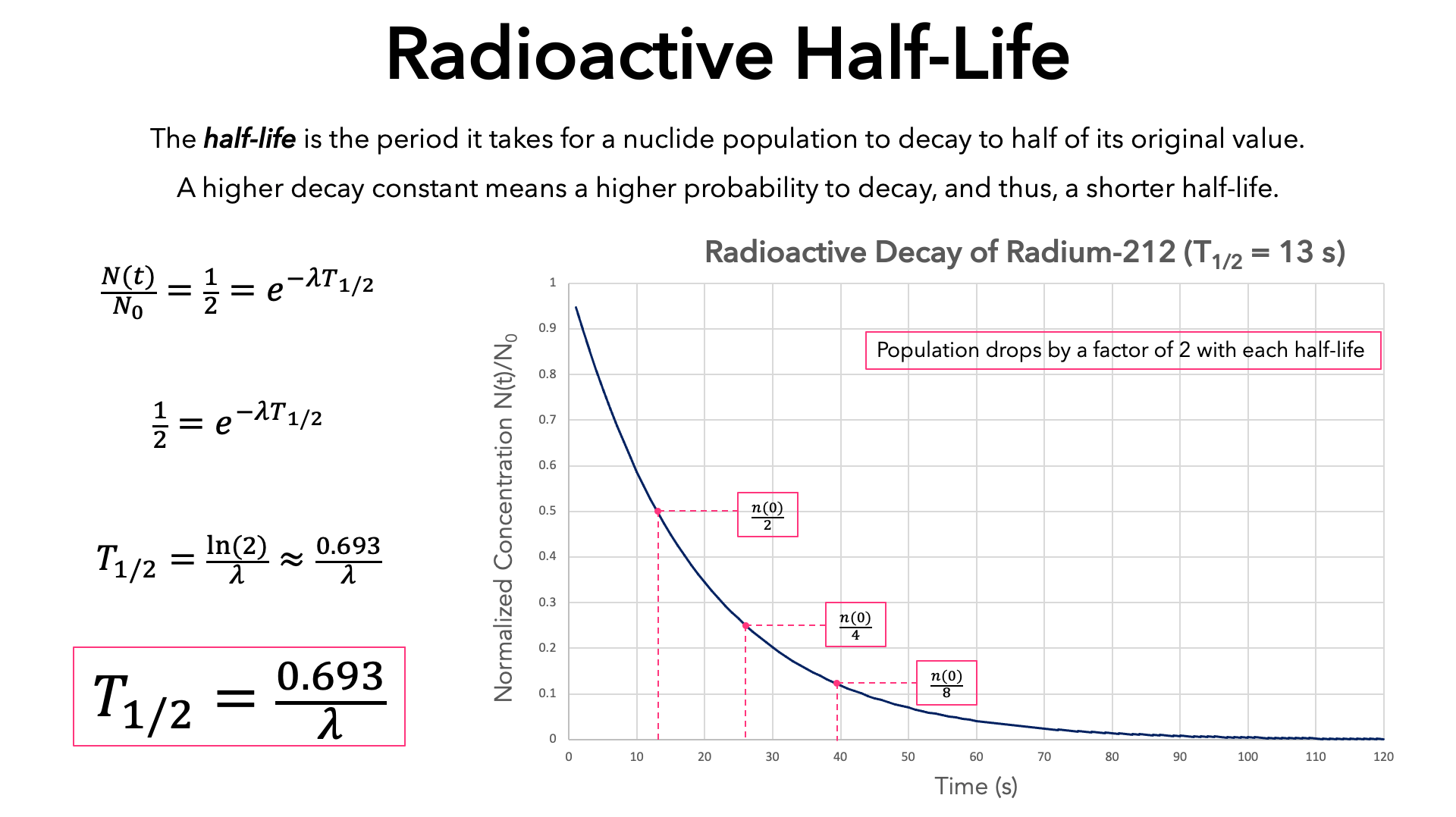
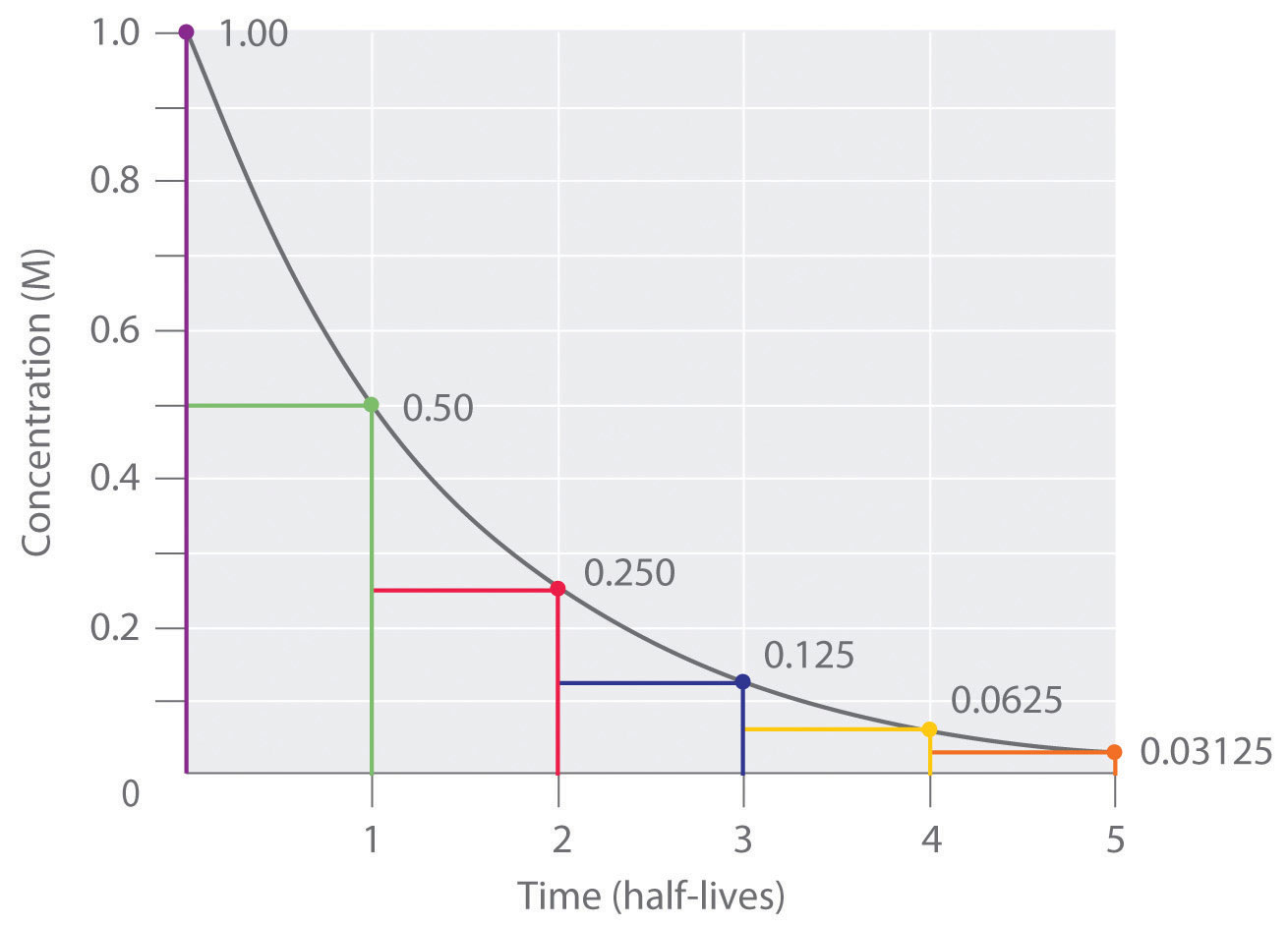
Nuclear Chemistry: Half-Life. Half Life Definition = time it takes for half of a radioactive sample to decay; describes rate of decay The stability of. - ppt download

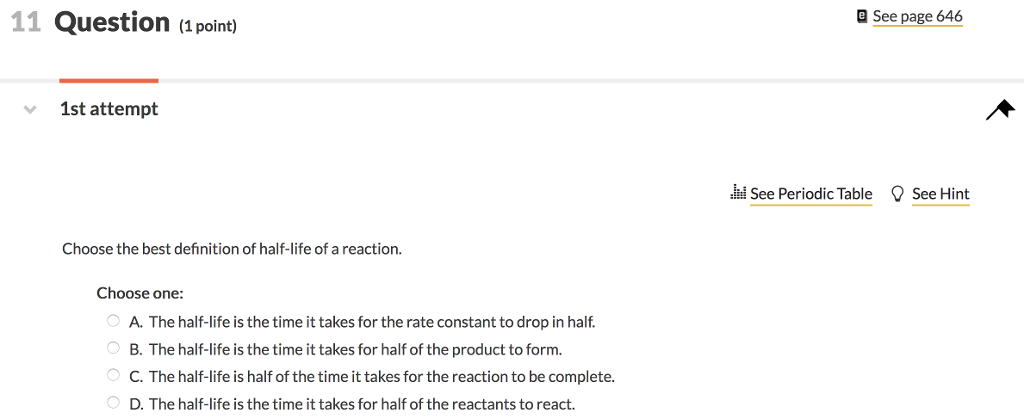
Define half life of a reaction Derive the relationship between half life and rate constant for a first Order - Chemistry - - 16068871 | Meritnation.com

The time for half - life period of a certain reaction A→ products is 1 hour. When the initial concentration of the reactant A is 2.0 mol L ^-1 , how much

What is meant by half-life? - It's a Question of Physics - The Atomic Age - Linda Hall Library - Kansas City, MO

Define the following terms:(i) Pseudo first - order reaction(ii) Half - life period of reaction (t1/2) .
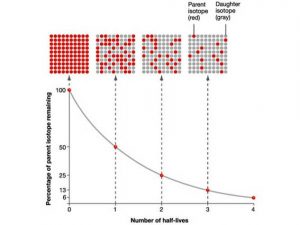
7.12 know the definition of the term half-life and understand that it is different for different radioactive isotopes - TutorMyself Chemistry