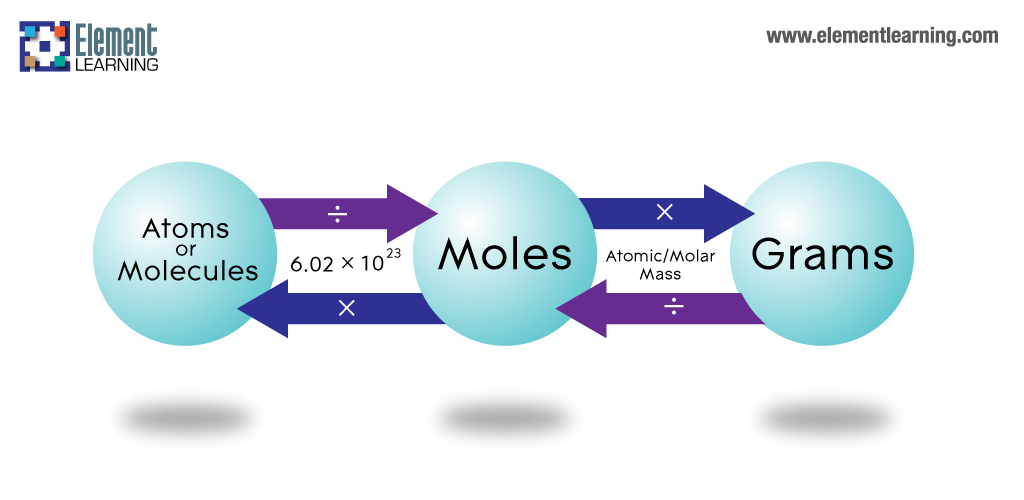
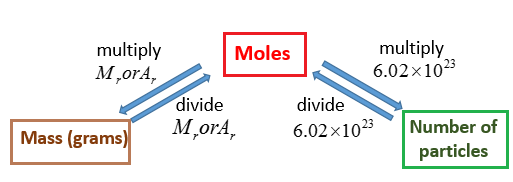
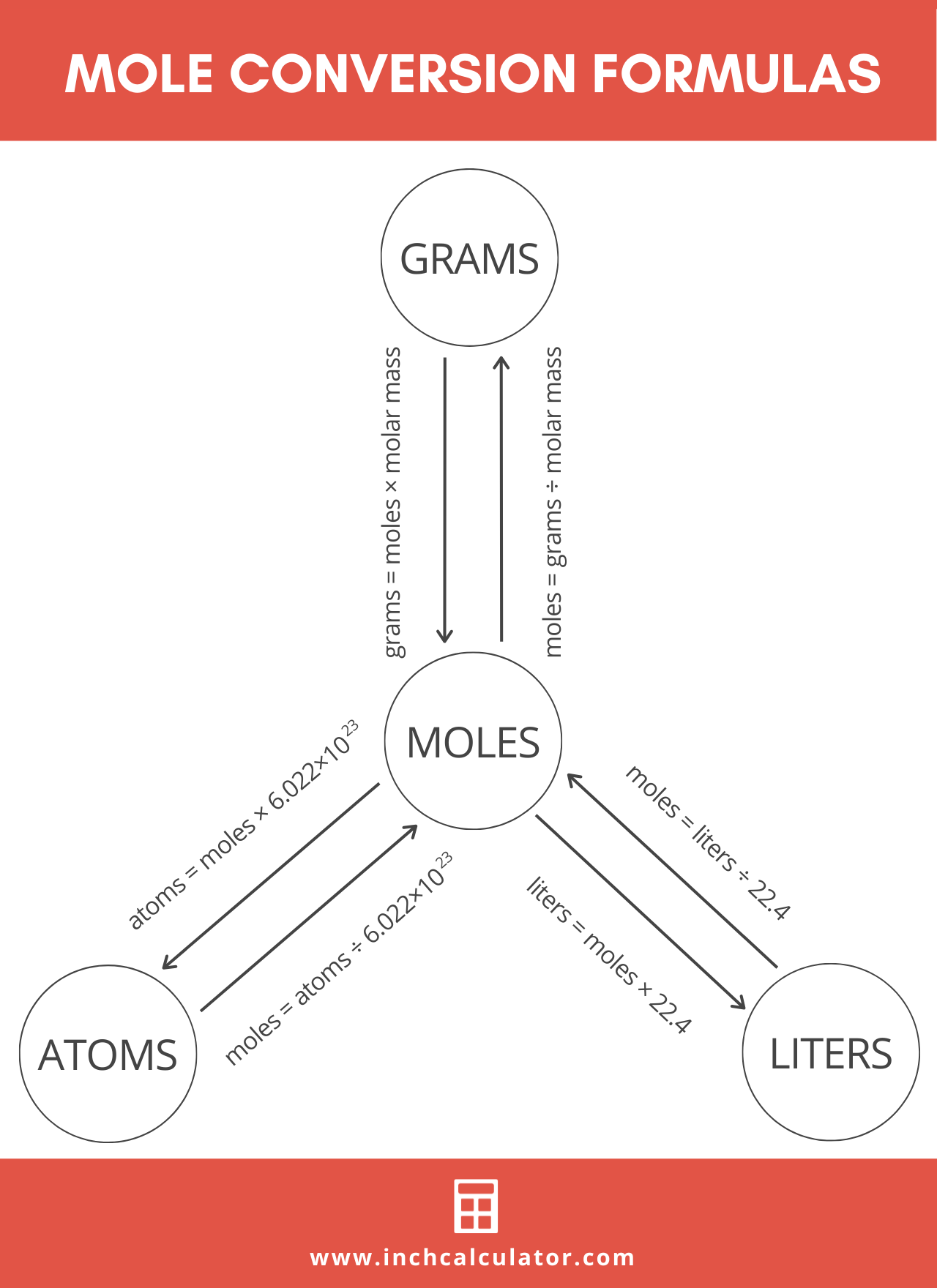
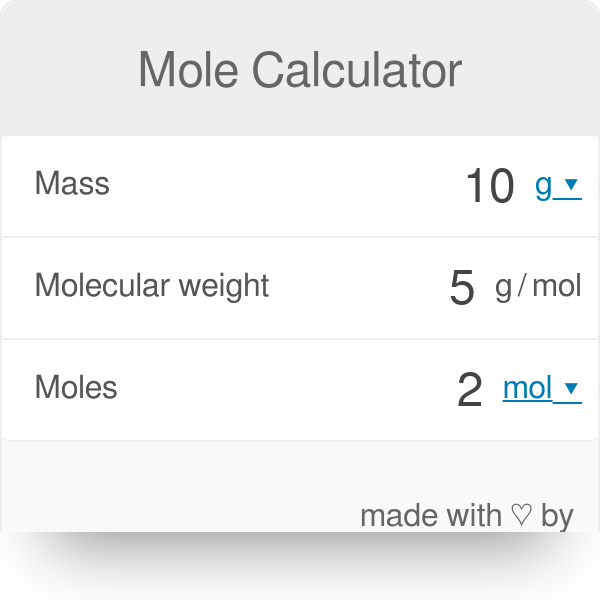
The Mole Calculating Formula/Molar Mass Calculate the molar mass of carbon dioxide, CO g + 2(16.00 g) = g One mole of CO 2 (6.02 x ppt download

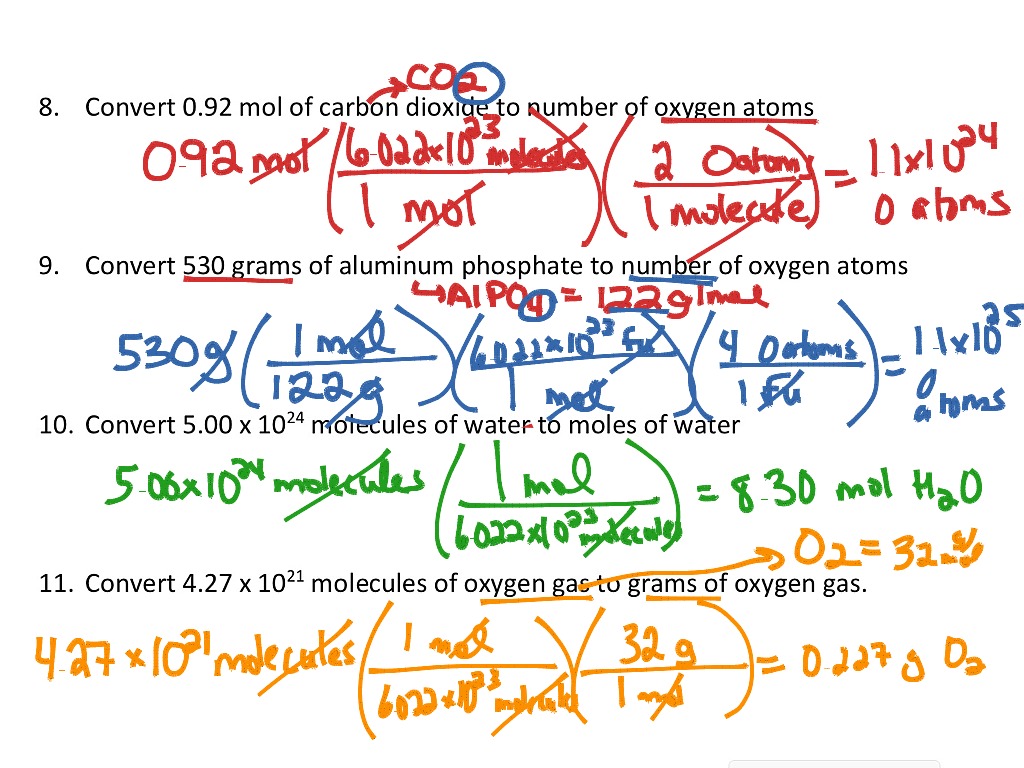
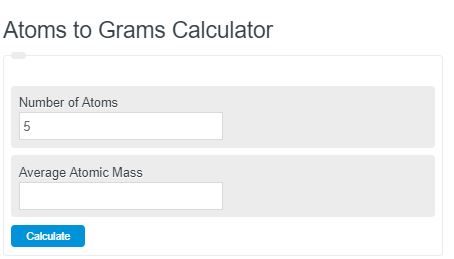
SOLVED: convert to grams: 2.53 X 1023atoms Fe mole Fe 55.85 grams Fe mole Fe 23.5 grams Fe 6.02 X 1023 atoms Fe Notice in these problems that we use Avogadros number