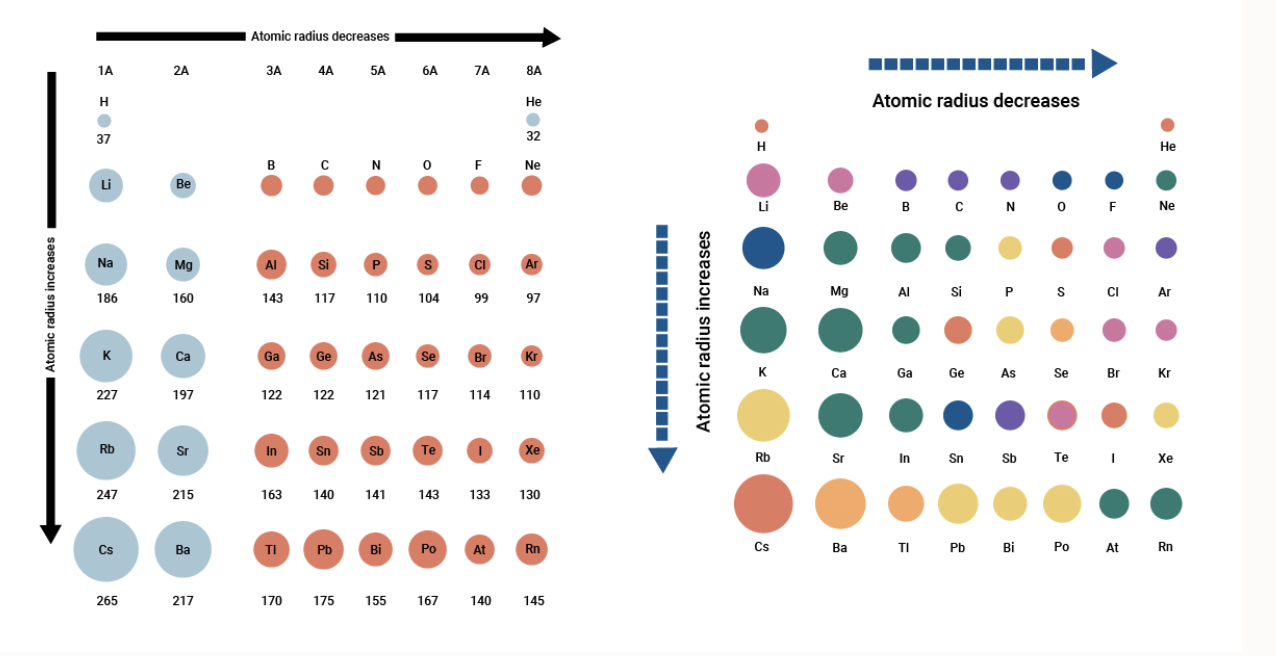
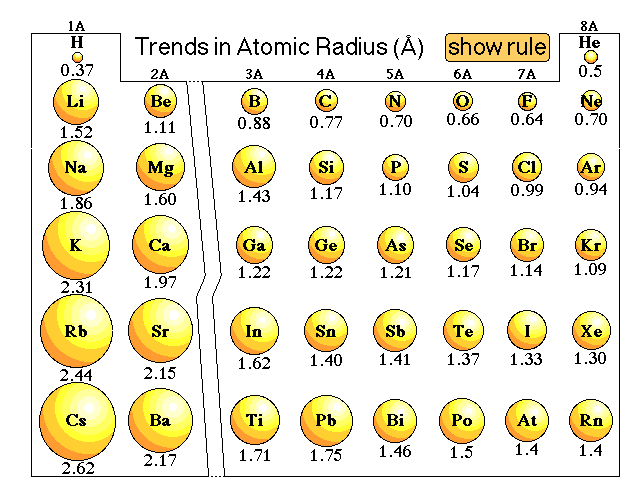
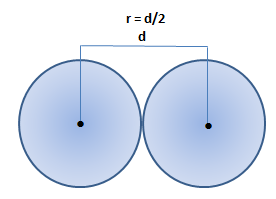
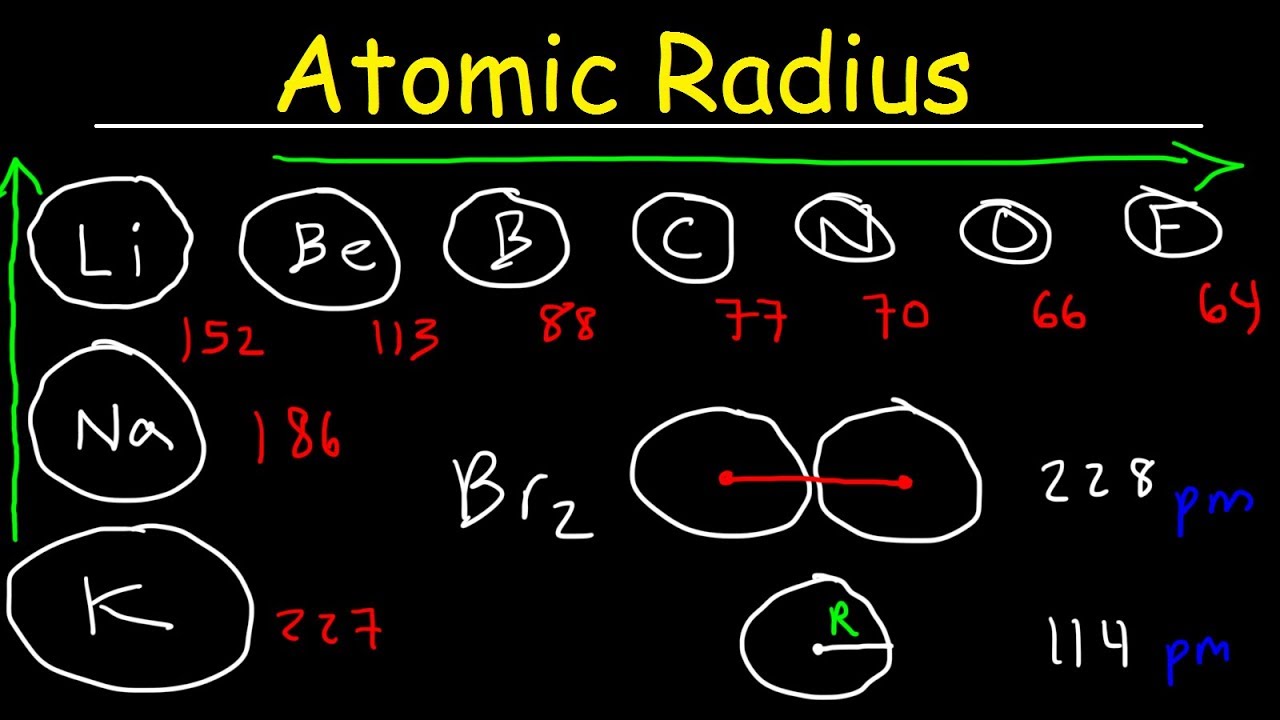
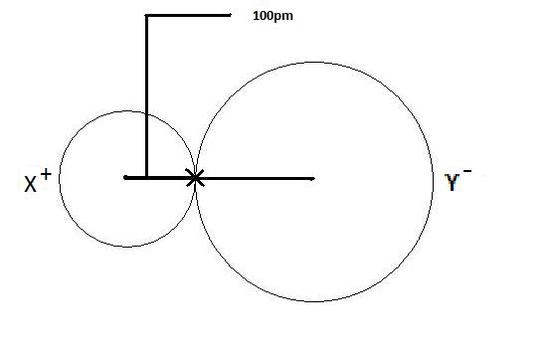
Gold (atomic mass = 197 u) has atomic radius = 0.144 nm. It crystallises in face centred unit cell. Calculate the density of gold. (No = 6.022xx10^(23)mol^(-1))

Lithium metal crystallizes in a body - centered cubic crystal. If the length of the side of the unit cell of lithium is 351 pm, the atomic radius of lithium will be:

Niobium has a density of 8.57 g/cm3 and crystallizes with the body-centered cubic unit cell. Calculate the radius of a niobium atom - Chemistry Stack Exchange
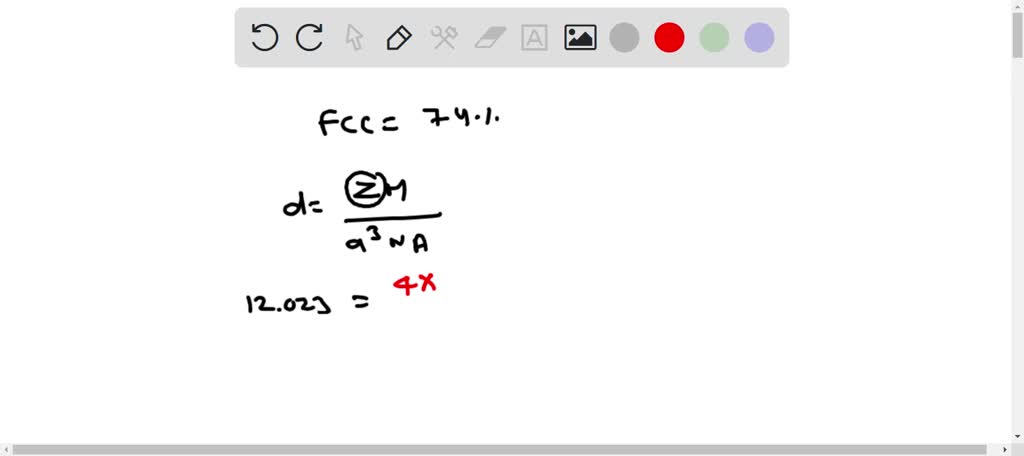
SOLVED: Palladium (at. wt. = 106) crystallizes in a face-centered cubic unit cell. Its density is 12.023 g/cm3 . Calculate the atomic radius of palladium and its packing efficiency.

Ionic Radius Trends, Basic Introduction, Periodic Table, Sizes of Isoelectric Ions, Chemistry - YouTube