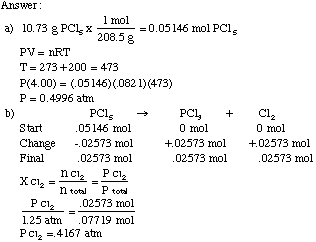
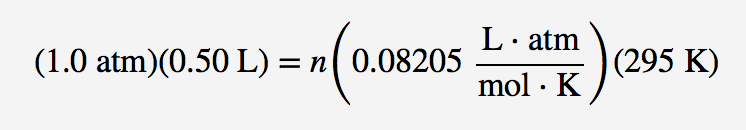If I have 21 moles of gas held at a pressure of 3800 torr and a temperature of 627°C what is the volume of the gas? | Socratic

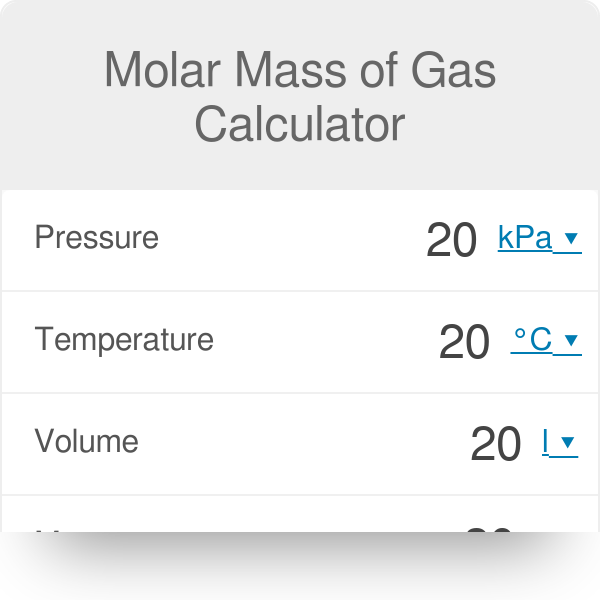
Ideal Gas Law Calculator (Pressure–Volume–Temperature–Amount) • Thermodynamics — Heat • Online Unit Converters

An ideal gas at 0.80 atmospheres and 87°C occupies 0.450 liter. How many moles are in the sample? - YouTube
Five moles of ideal gas expand isothermally and reversibly from pressure 10 ATM to 2 ATM at 300 K. What is the largest mass which can be lifted through a height of

molar gas volume Avogadro's Law moles and mass calculations gcse chemistry calculations igcse KS4 science A level GCE AS A2 O Level practice questions exercises

Question Video: Calculating 𝐾_𝑝 at Equilibrium for a Mixture of Nitrogen, Hydrogen, and Ammonia | Nagwa

OpenStax College Physics Solution, Chapter 13, Problem 28 (Problems & Exercises) | OpenStax College Physics Answers